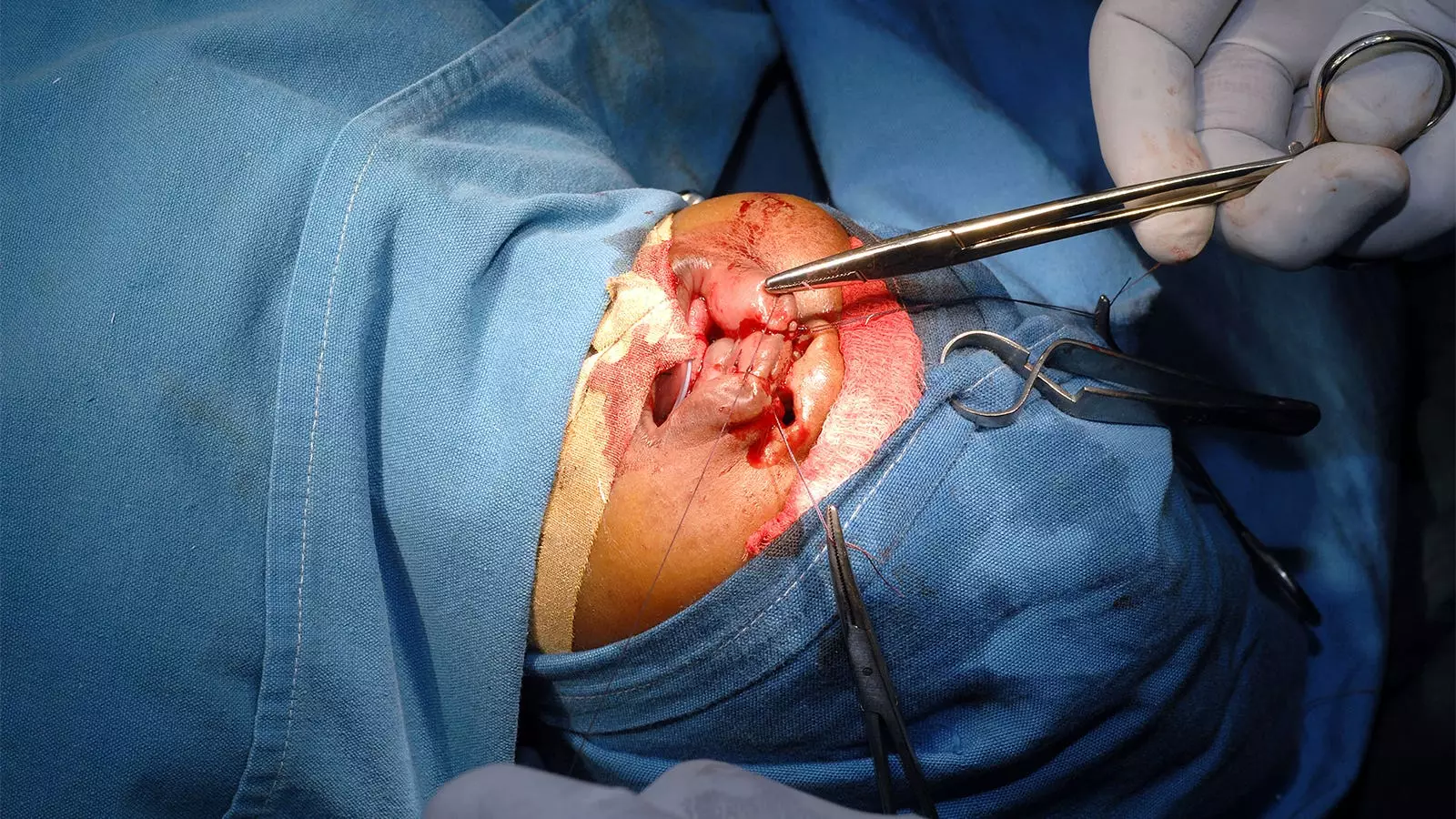
In the TOPS clinical trial, researchers conducted a study to determine the effects of primary surgery timing on velopharyngeal function in infants with isolated cleft palate. The trial included 558 infants in Europe and South America, comparing outcomes between those who had surgery at 6 months and those who had surgery at 12 months. The results showed that infants who had surgery at 6 months had a lower risk of insufficient velopharyngeal function later in childhood compared to the 12-month surgery group. However, a critical analysis of the study reveals certain limitations and factors that should be considered when interpreting the findings.
The Importance of Timing
The study aimed to address the association between later repair and poorer speech outcomes, which has been suggested in previous studies. Timing of primary surgery has long been a topic of debate in the medical community, with some advocating for earlier intervention and others cautioning against potential risks. The TOPS trial sought to provide a definitive answer through a randomized controlled trial.
The findings of the TOPS trial should be interpreted in the context of the overall trial design. The study showed that the 6-month surgery group had a lower risk of insufficient velopharyngeal function at age 5, but there were no clear differences in other secondary outcomes. It is important to note that speech evaluations were conducted without considering additional interventions such as speech therapy or secondary surgery. This could potentially impact the interpretation of the results.
Potential Confounding Factors
One potential confounding factor is the occurrence of secondary surgery to treat velopharyngeal insufficiency. The study found that more children in the 6-month surgery group required secondary surgery for this issue. This raises the question of whether the better primary outcome response in the 6-month group was solely due to the timing of surgery or if it was influenced by the increased occurrence of secondary surgery. Without standardization of the timing of secondary surgery, the trial is susceptible to treatment biases.
The TOPS trial had certain limitations that should be considered. Speech evaluations were conducted using a velopharyngeal composite score, which may not capture the full complexity of speech quality. In addition, the study relied on speech recordings, photographs, and impressions, which may introduce variability in the assessment process. The study was also underpowered to detect differences in safety between the two groups.
The TOPS clinical trial provides valuable insights into the effects of primary surgery timing on velopharyngeal function in infants with isolated cleft palate. The findings suggest that surgery at 6 months may lead to better outcomes in terms of velopharyngeal function at age 5. However, it is important to critically analyze the study and consider limitations such as the potential confounding factors and the variability in speech evaluations. Further research is needed to fully understand the relationship between primary surgery timing and long-term speech outcomes in this population.

Leave a Reply